Navigating the Gray Area Between HER2 Low and HER2 Negative Status
2026-03-20
By admin
The New Frontier in Breast Cancer Diagnostics
For many years, HER2 testing in breast cancer followed a clear-cut rule. A tumor was either HER2-positive, which meant the patient could receive HER2-targeted drugs, or HER2-negative. That simple division changed with the arrival of the HER2-low category. New antibody-drug conjugates (ADCs) have demonstrated strong results in tumors that show only small amounts of the HER2 protein on cell surfaces.

This change creates a difficult spot for pathologists and diagnostic labs. The difference between an IHC score of 0 (negative) and an IHC score of 1+ (low) now carries major weight. It decides whether a patient qualifies for treatments that can extend life. Celnovte supplies a set of advanced immunohistochemistry tools built to handle the fine distinctions demanded in today’s precision oncology work.
Understanding the Gray Area: HER2-Low vs. HER2-Negative
The Clinical Shift and the Role of ADCs
HER2-low includes tumors scored as IHC 1+, or as IHC 2+ when in situ hybridization (ISH) gives a negative result. Studies show that roughly half of the cases once labeled HER2-negative actually belong in the HER2-low group. Finding these patients matters a great deal. ADCs are designed to seek out cells with low HER2 levels, carry powerful drugs straight to the cancer, and leave normal tissue mostly untouched.
The Challenge of Reproducibility in IHC Scoring
The greatest difficulty in this borderline zone lies in the subjective nature of reading faint staining by eye. Telling apart very weak, partial membrane staining (IHC 1+) from complete absence of stain (IHC 0) takes skilled pathologists and tightly controlled staining methods. Small differences in how tissue is fixed, how strongly the antibody binds, or how well the detection reagents work can shift the final score. Such shifts sometimes prevent patients from receiving ADC treatment that could help them.
Celnovte’s Solutions for High-Precision HER2 Testing
HER2 Primary Antibodies: Superior Sensitivity for Low-Expression Detection
Accurate HER2-low calls start with a reliable primary antibody. Celnovte provides a selection of carefully produced HER2 monoclonal antibodies (RMab/MMab) that stand out for their ability to detect low-level expression.
-
Core Advantage: Within our full antibody lineup, 41 self-developed clones received “Optimal” or “Good” ratings from NordiQC, the respected external quality assessment program.
-
Specific Benefit: These antibodies generate sharp, distinct membrane staining even when the HER2 protein is present in small quantities. That clarity cuts down on cases wrongly called IHC 0.
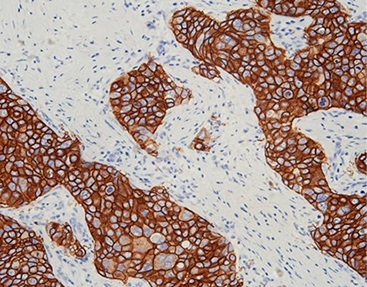
MicroStacker™ Detection System: Enhancing Signal Without the Noise
High detection strength loses value if it also picks up unwanted background color. Celnovte developed the MicroStacker™ IHC Detection System to address exactly that issue.
-
Technology Detail: Conventional HRP polymer methods rely on large dextran carriers. MicroStacker™ instead uses a small, tightly packed micro-polymer structure. The design allows precise layering of peroxidase enzymes and antibody fragments.
-
Result: The arrangement delivers outstanding signal strength while keeping non-specific staining very low. It avoids problems linked to endogenous biotin. In borderline cases, this clean, high-contrast picture helps confirm true IHC 1+ staining.
CNT 360 Fully Automated Stainer: Eliminating Manual Variability
Consistent results across labs depend on removing steps that vary from person to person. The CNT 360 Full Automatic IHC & ISH Stainer serves as Celnovte’s main platform for reliable, high-volume work.
-
Features: It processes up to 60 slides at once and finishes a complete run in about 2.5 hours. The instrument runs several different protocols on the same deck without switching hardware.
-
Impact: Because it controls reagent drops, timing, and temperature automatically, the CNT 360 removes many sources of human variation. Slides receive identical treatment every time. Large pathology centers gain steady quality that supports fast, dependable reporting.

Application Scenarios: Where Precision Meets Clinical Practice
Accurate Identification of Candidates for ADC Therapy
-
The Problem: A lab receives a metastatic breast cancer biopsy with an earlier report that called the HER2 result borderline—somewhere between IHC 0 and 1+.
-
The Solution: The lab runs the sample again with Celnovte’s HER2 primary antibody paired with the MicroStacker™ detection reagents.
-
Application: The sensitive combination picks up faint, incomplete membrane staining in more than 10% of tumor cells. This finding shifts the classification to HER2-low and opens the door to an ADC regimen that the patient would have missed otherwise.
High-Throughput Standardization in Large-Scale Pathology Labs
-
The Problem: A busy regional lab handles hundreds of breast cancer specimens each day. Keeping stain quality the same across multiple technologists and work shifts proves hard with manual or older semi-automatic equipment.
-
The Solution: The lab adopts the CNT 360 Fully Automated Stainer for its HER2 workflow.
-
Application: With NMPA, GMP, and CE IVDR certifications in place, the system delivers uniform staining day after day. Retesting drops sharply, turnaround time shortens, and clinicians start appropriate therapy sooner.
Conclusion: Redefining the Standard of Care
The line separating HER2-low from HER2-negative has become a key factor in choosing breast cancer treatment. Diagnostic tools must match the accuracy that modern therapies require. Celnovte, based in Rockville, MD, and active in more than 40 countries, focuses on meeting that need. Our advanced polymer chemistry, combined with full automation, helps pathologists move through uncertain cases with solid confidence.
Call to Action
Raise the accuracy of your laboratory’s HER2 testing today. Review Celnovte’s NordiQC-rated antibodies and automated IHC platforms. Reach our team at info@celnovte.com or explore the Solution Center to see how we can support your HER2-low diagnostic process.
FAQ
Q: Why is IHC 1+ vs. IHC 0 so critical in modern pathology?
A: Following approval of ADCs such as trastuzumab deruxtecan, patients whose tumors score IHC 1+ (HER2-low) now qualify for targeted drugs that used to be limited to IHC 3+ (HER2-positive) tumors. An IHC 0 score rules them out.
Q: How does the “detection system” affect HER2-low scoring?
A: Older detection methods sometimes fail to show very low HER2 levels or add so much background that weak staining disappears. Polymer-based systems built for high sensitivity capture the lower range of expression more reliably.
Q: Is ISH testing still required for HER2-low cases?
A: Yes, but only for IHC 2+ results. Current guidelines require ISH confirmation on IHC 2+ samples. If ISH is negative, the tumor counts as HER2-low. IHC 1+ cases qualify as HER2-low without ISH.
Q: Can automation truly improve the reproducibility of HER2 scoring?
A: Yes. Automated systems hold every step—antigen retrieval heat, reagent contact time, wash cycles—constant. That consistency reduces differences within the same lab and between different labs, which often cause conflicting reads in borderline situations.



