A Guide to Hormone Receptor Status What Happens If ER Is Positive
2026-03-05
By admin
Getting a pathology report after a biopsy stands as a key moment for any patient and doctor. Among the different biomarkers shown, few match the importance of hormone receptor status. If a report shows that a tumor is ER positive (Estrogen Receptor positive), it starts a focused clinical path. But what does this reveal about the biology, and why does the reliability of this particular test matter so much for treatment results? This guide, brought to you by Celnovte, will help you explore the crucial aspects of ER testing in breast cancer.
Understanding Hormone Receptor Status in Breast Cancer
Hormone receptor status acts as a simple method to explain whether breast cancer cells have receptors that bind to estrogen and progesterone. These hormones connect to the receptors. They provide the energy that signals cancer cells to multiply and divide.
What Does “ER Positive” Actually Mean
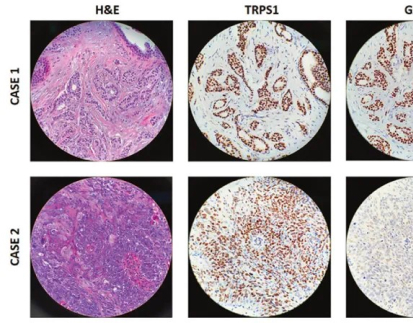
An ER-positive diagnosis means cancer cells contain a high amount of receptors for estrogen. Doctors determine this in practice using Immunohistochemistry (IHC). When 1% or more of the tested cells show staining for these receptors, experts classify the tumor as ER-positive. This fact defines the tumor’s character. It also directs the whole treatment plan.
The Biological Mechanism of Estrogen Receptors
Estrogen receptors are proteins located within the cells. When estrogen moves through the blood, it enters the cell and connects to these receptors. In ER-positive breast cancer, this connection sets off a series of genetic messages that speed up the cell cycle. Based on medical study findings, around 70% to 80% of all breast cancers are ER-positive. This makes it the most frequent type of illness. Since the growth depends on hormones, it creates a clear weak spot that doctors can target with specific treatments.
Clinical Implications: What Happens if the ER is Positive
An ER-positive outcome often counts as a good sign when it comes to treatment choices. It allows access to therapies that cause far less harm than standard chemotherapy, yet they work well at stopping the cancer from coming back.
Targeted Therapy and Treatment Pathways
If a patient shows ER positive, the main plan involves Endocrine Therapy (also called hormone therapy). The aim is to reduce estrogen levels in the body or prevent estrogen from reaching the cancer cells. Typical options include:
- Selective Estrogen Receptor Modulators (SERMs): Like Tamoxifen, which occupy the receptors and stop estrogen from attaching.
- Aromatase Inhibitors (AIs): These block the body from making estrogen in women after menopause.
- Ovarian Suppression: For women before menopause, this halts hormone production in the ovaries.
Prognosis and Long-term Outcomes
In terms of numbers, ER-positive cancers usually grow at a slower pace than ER-negative types. Research from clinics shows that patients with hormone-receptor-positive tumors tend to have a better short-term prognosis. Yet, since these cells can stay inactive for many years, ongoing checks remain vital. The effectiveness of these long-range treatment strategies depends fully on the starting IHC test delivering complete accuracy.
The Critical Need for Diagnostic Precision
The choice to place a patient on hormone therapy for five to ten years hinges on one IHC slide. A wrong negative might keep a patient from vital targeted care, while a wrong positive could bring unneeded treatment and unwanted effects.
Challenges in Hormone Receptor IHC Staining
IHC staining for ER reacts to several factors, such as how long the tissue stays fixed and the standard of the primary antibody applied. Uneven staining might cause spotty outcomes or extra noise that hides the real receptor numbers. This explains why pathology labs need to use reagents that deliver top levels of sensitivity and exactness.
The Significance of NordiQC Assessment for Labs
NordiQC (Nordic Immunohistochemical Quality Control) acts as an independent global group that reviews antibody performance in pathology. For a producer, earning an “Optimal” score from NordiQC proves top quality. Celnovte has shown this high standard time and again, with its ER primary antibodies gaining the optimal NordiQC assessment for 6 consecutive years.
Celnovte’s High-Performance Solutions for ER Testing
As a focused maker of pathological diagnostic reagents, Celnovte supplies a full range of products aimed at removing doubt in diagnostics. Our ER testing tools gain trust from more than 2,300 leading hospitals in China and reach over 40 countries around the world.
Celnovte ER Primary Antibody (Rabbit Monoclonal)
The Celnovte ER Primary Antibody comes as a self-cloned rabbit monoclonal antibody (RMab) built for spotting Estrogen Receptor alpha in a qualitative way.
- Features: It provides outstanding signal-to-noise ratios along with clear nuclear staining.
- Advantages: Different from certain polyclonal antibodies, our RMab brings strong binding and steady results across various tissue groups. It holds the Class III NMPA certificate, meeting the toughest clinical rules for diagnostic work.
Celnovte PR Primary Antibody (Companion Diagnostic)
Since ER and PR (Progesterone Receptor) status often get checked together to form a complete “Hormone Receptor” profile, Celnovte provides a strong-binding PR Primary Antibody.
- Details: Similar to the ER antibody, our PR clone has also earned optimal NordiQC assessments.
- Benefit: Applying a paired set of ER and PR antibodies from the same maker supports uniform staining methods and simpler reading for the pathologist.
MicroStacker™ Polymer Detection Technology
The final slide’s quality relies not only on the antibody but also on the detection setup. Celnovte’s MicroStacker™ technology changes the game for ER/PR testing.
- Mechanism: It uses a stacked layering of F(ab’) fragments from secondary antibodies and enzymes on a micro-polymer base.
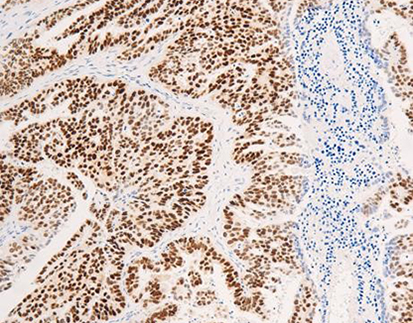
- Why it Matters: This design stays more compact and responsive than old-style large dextran-based polymers. It skips biotin, which cuts out non-specific background staining from natural biotin in the tissue. As a result, it produces the sharp nuclear staining needed for figuring out the Allred score or the share of ER positivity.
About Celnovte: Elevating Precision in Pathology
Celnovte focuses on research, development, and production of advanced pathological diagnostic reagents and tools. Based in Rockville, Maryland, for reagent and instrument work, we run NMPA and GMP-approved production sites in China. Our goal is to boost accuracy in cancer diagnostics through new ideas, backed by approvals like ISO13485, ISO9001, FDA, and CE IVDR. With more than 120 self-cloned primary antibodies, we commit to aiding labs in making better choices, doing stronger work, and becoming more effective for patients globally.
Conclusion
When ER turns positive, it points to a controllable and directed route ahead for breast cancer care. Still, the trustworthiness of that “positive” finding matches only the tools used in the lab. By picking Celnovte’s NordiQC-optimal antibodies and our cutting-edge MicroStacker™ detection systems, pathologists can give oncology groups the full trust required to assign vital endocrine therapies. Accuracy in the lab turns straight into hope in the clinic.
FAQ
Q: Can a tumor’s ER status change over time?
A: Yes, receptors can shift during recurrence or spread, so doctors frequently re-biopsy new tumors to verify hormone status.
Q: What is the difference between ER and PR?
A: ER reacts to estrogen, while PR reacts to progesterone. Most ER-positive cancers also show PR-positive, and having both typically means a stronger response to hormone therapy.
Q: Why is the Allred score used in ER testing?
A: The Allred score mixes the percentage of positive cells with the strength of the staining to create a total score (0-8). This helps doctors forecast how well hormone therapy might work.
Q: Does Celnovte provide automated solutions for ER staining?
A: Yes, Celnovte supplies various fully automated IHC slide stainers and ready-to-use (RTU) reagents to deliver consistent and repeatable staining outcomes in all lab processes.






![[Instrument] MicroStacker™ Plus Polymer Detection Kit [Instrument] MicroStacker™ Plus Polymer Detection Kit](https://www.celnovte.com/wp-content/uploads/2020/08/3.gif)